Galvanic Corrosion Cable Cleats CMP Products Limited
Galvanic reaction is the principle upon which batteries are based. See the table of standard electrode potentials for more details. Galvanic series (most noble at top) The following is the galvanic series for stagnant (that is, low oxygen content) seawater. The order may change in different environments. [1] Graphite Palladium Platinum Gold Silver

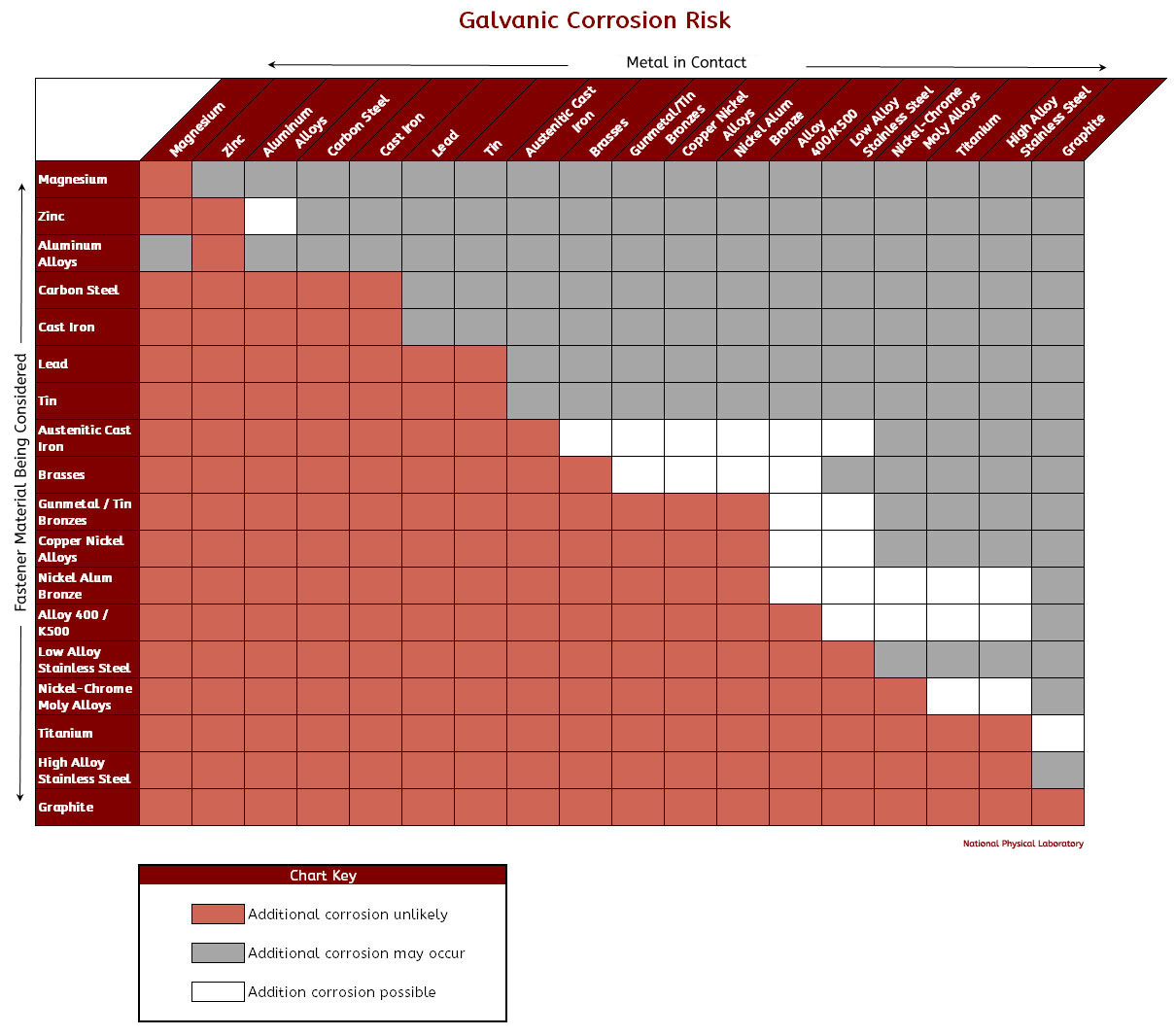
Galvanic Reaction Chart All Points Fasteners
The galvanic series determines the electrochemical potential and nobility of metals and metal alloys. Corrosivity Each alloy or metal has a distinctive corrosion potential. The more negative a metal or alloy is, the more likely it is to suffer galvanic corrosion.

The Galvanic Series the essential guide EngineeringClicks
Galvanic series relationships are useful as a guide for selecting metals to be joined, will help the selection of metals having minimal tendency to interact galvanically, or will indicate the need or degree of protection to be applied to lessen the expected potential interactions.
Galvanic Chemistry Dictionary & Glossary
Simply speaking, galvanic corrosion is the damage or deterioration of metal that takes place between dissimilar metals because of an electrochemical reaction.
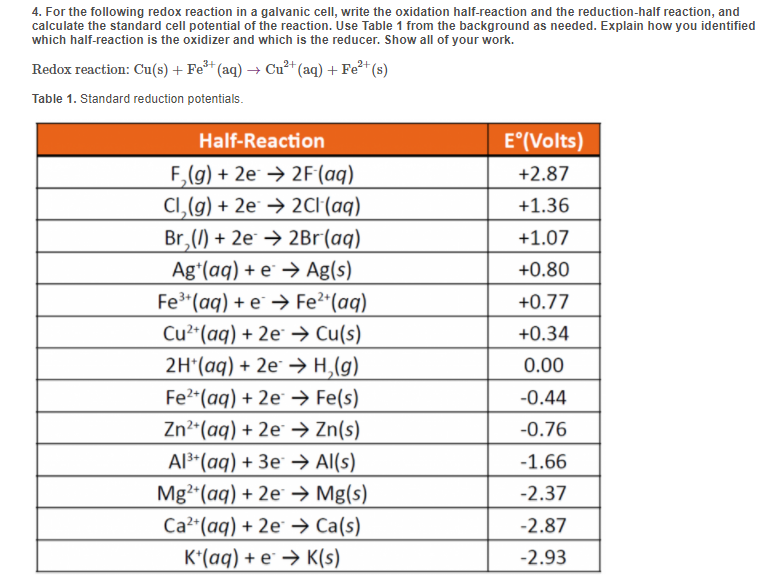
Solved 4. For the following redox reaction in a galvanic
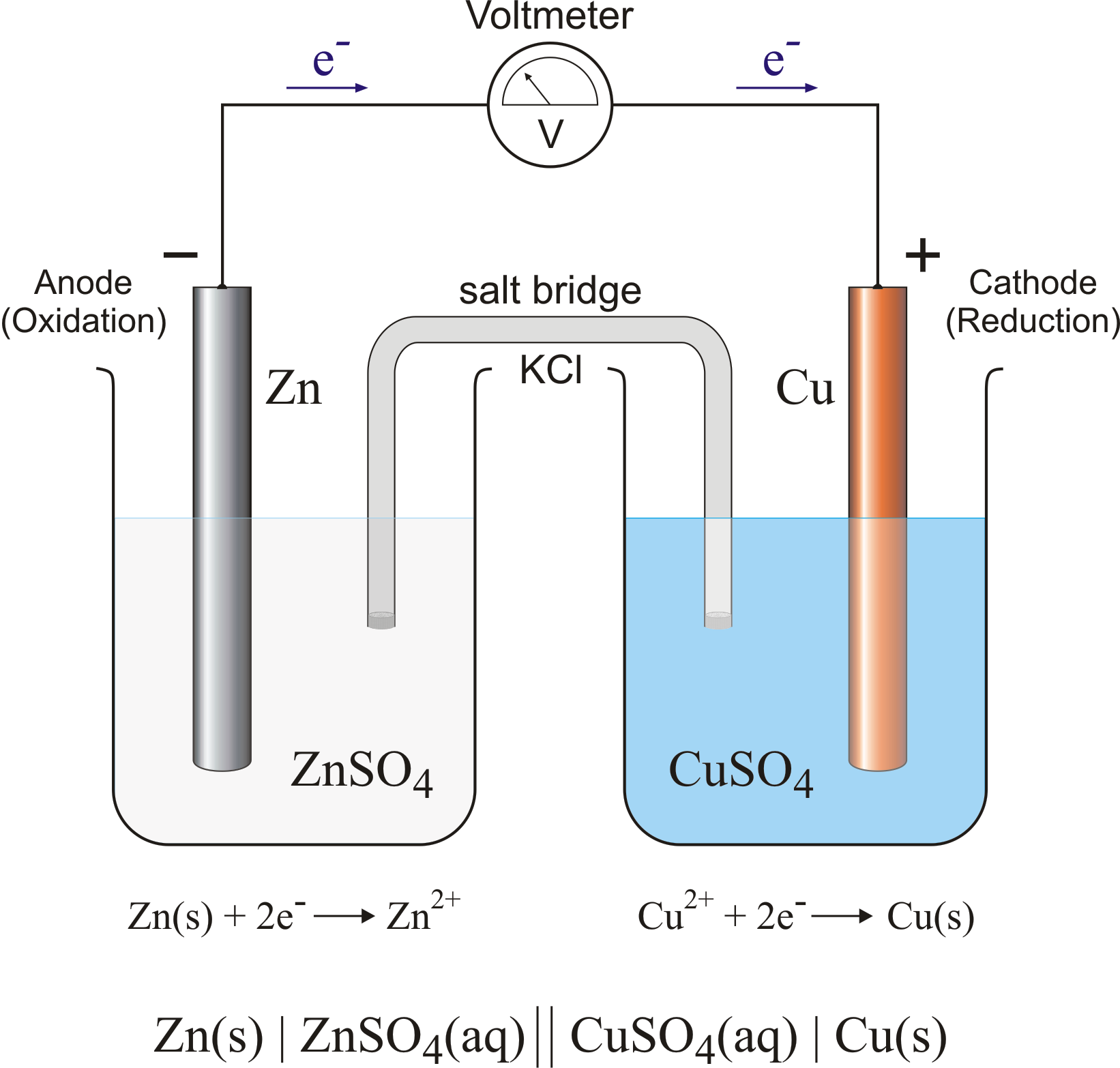
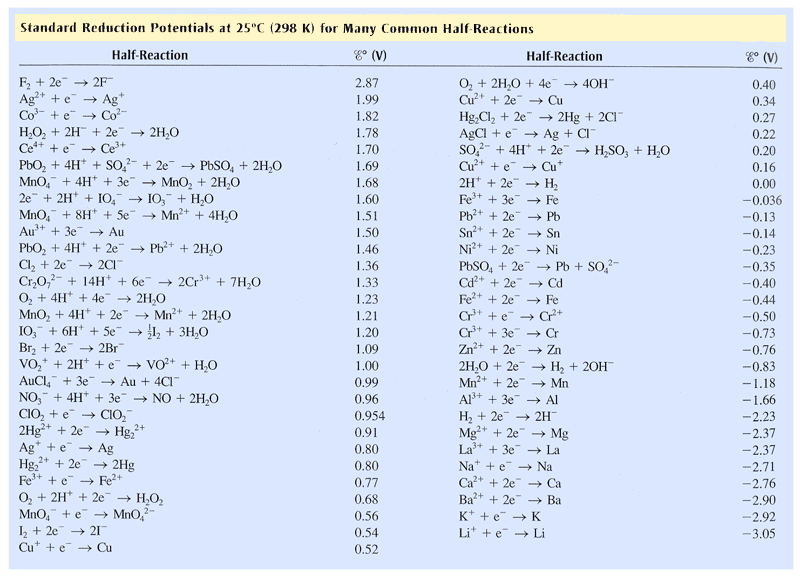
Corrosion potential and the galvanic series. When a metal corrodes in an electrolyte, atoms from the metal separate into ions and electrons (e - ), with the ions dissolving into the electrolyte. For example, for iron (Fe) the reaction is: Fe → Fe 2+ + 2e −. This is called an anodic reaction and Fe 2+ ions are formed.
Galvanic potential of metals after chromate conversion coating
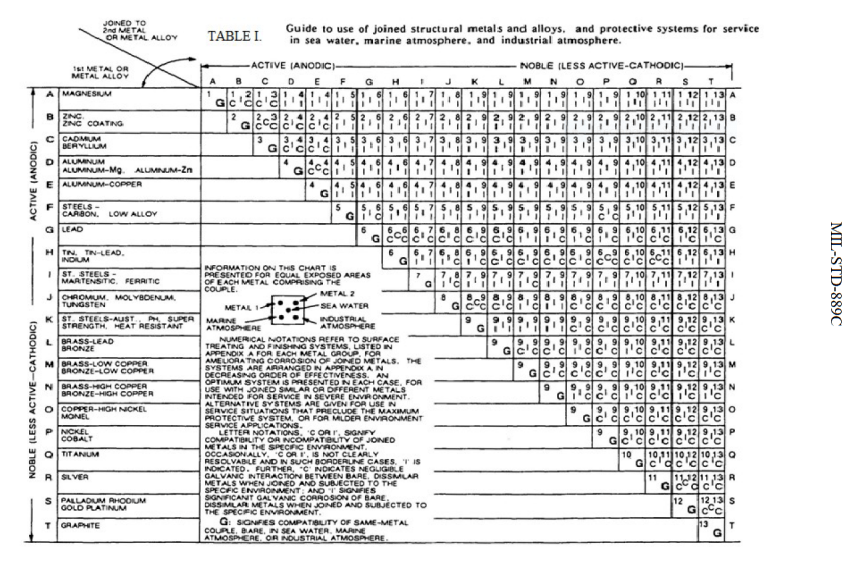
up a galvanic action which results in the deterioration of one of them. The following is a list of the more common commercial metals, sequenced according to what is known as the "Galvanic Table": THE GALVANIC TABLE 1. Aluminum 2. Zinc 3. Steel 4. Iron Anodic or Active (+) 5. Nickel 6. Stainless Steel Series 400 ↔ 7. Tin 8. Lead 9. Brass 10.
Galvanic Corrosion Chart
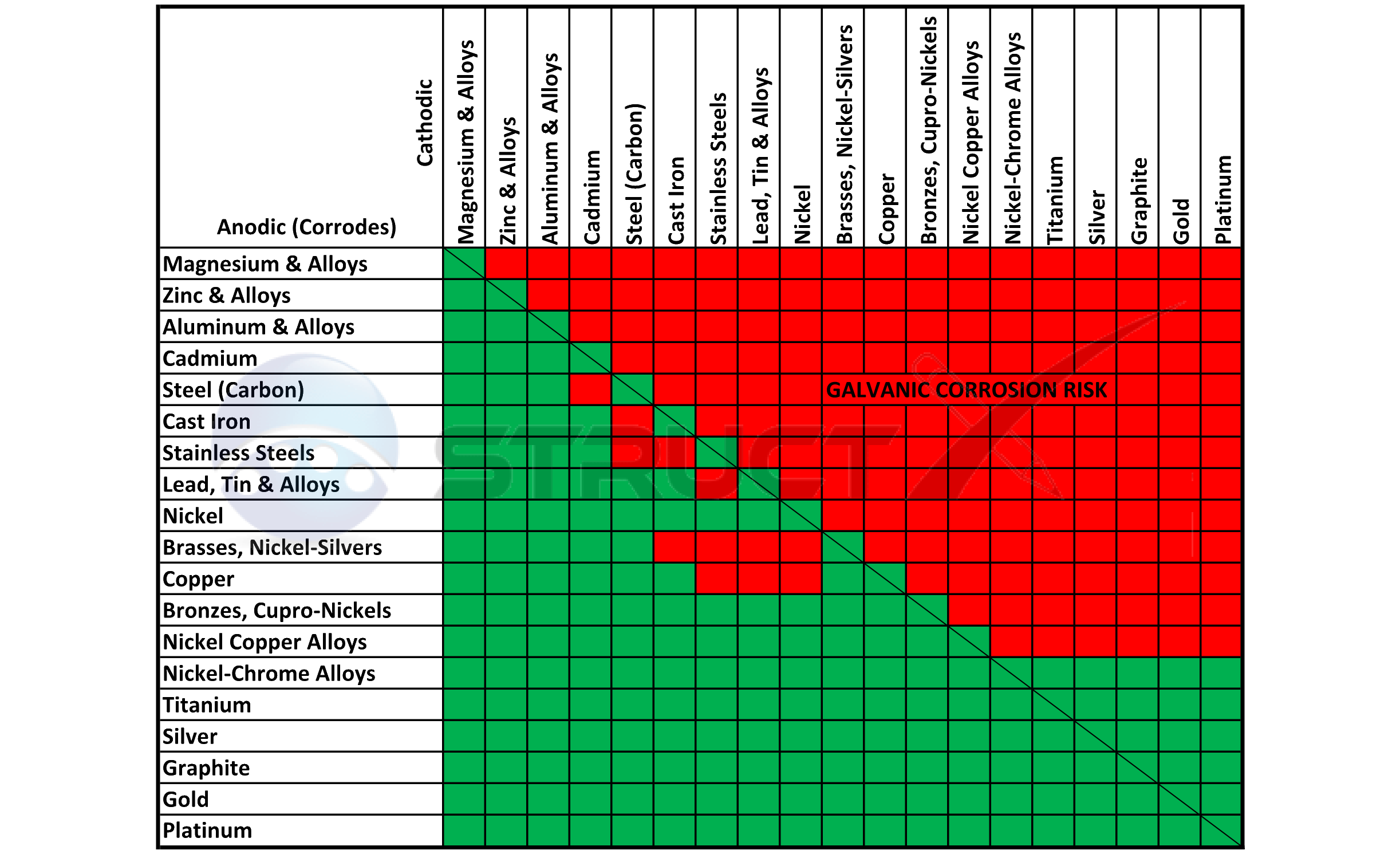
Galvanic Table The following galvanic table lists metals in the order of their relative activity in seawater environment. The list begins with the more active (anodic) metal and proceeds down the to the least active (cathodic) metal of the galvanic series.

Mixing Metals in Fasteners
75 of The Top 100 Retailers Can Be Found on eBay. Find Great Deals from the Top Retailers. eBay Is Here For You with Money Back Guarantee and Easy Return. Get Your Galvanic Today!

Separating Galvanic Metals JLC Online
Print. ResearchGate. (2014, March). Galvanic series of various materials in flowing seawater (2.5-4 m/s) at temperatures in the range from 5 to 30oC. [ upload Retrieved from https://www.researchgate.net/figure/Galvanic-series-of-various-materials-in-flowing-seawater-25-4-m-s-at-temperatures-in_fig1_260528991

Galvanic Corrosion Chart Pay attention! You might accidentally learn
Shop Like A Billionaire, Come & Check Everything At A Surprisingly Low Price. Come and check everything at a surprisingly low price, you'd never want to miss it.
Solved Consider The Following Galvanic Cells. For Each Ga...
Galvanic corrosion (also called bimetallic corrosion or dissimilar metal corrosion) is an electrochemical process in which one metal corrodes preferentially when it is in electrical contact with another, in the presence of an electrolyte.

Galvanic Corrosion A Guide for Architects (with a Galvanic Series Chart)
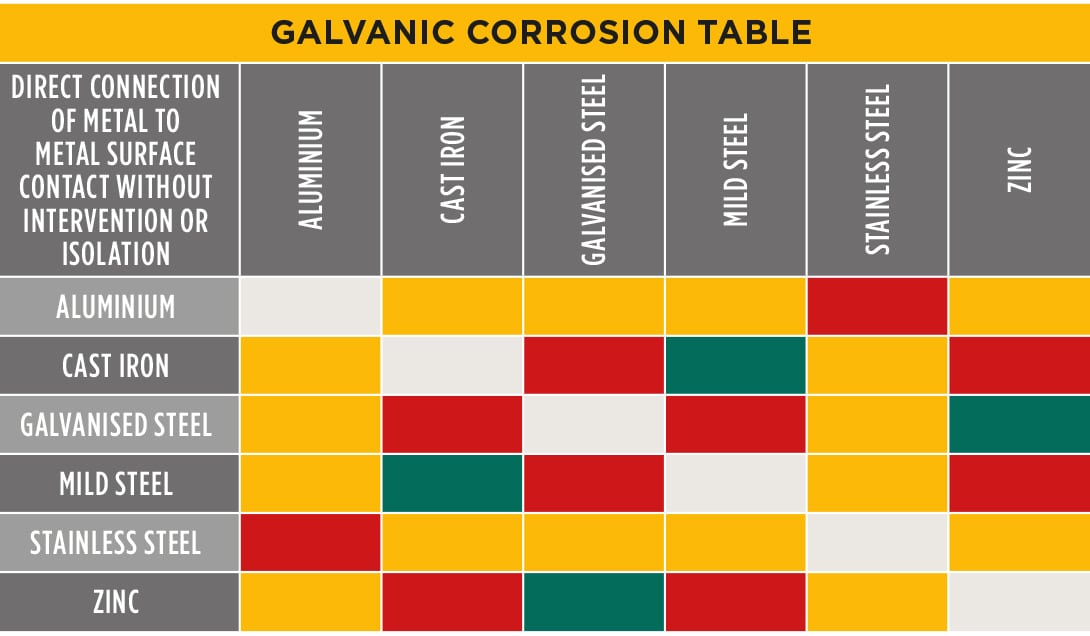
Below is a galvanic reaction chart for dissimilar metals. Please understand that green represents "lower risk" not "no risk." It should be noted that if sacrificial plating is incorporated in the fastener design, then galvanic action can result in the deterioration of the sacrificial coating, rather than of the fastener.

Galvanic Potential Chart
The Galvanic Series, also called the electro-potential series, lists metals in the order of their nobility. (Noble metals are those that are resistant to corrosion and oxidation.) When two metals are immersed in an electrolyte, while also being connected externally by a conductor, the less noble metal experiences galvanic corrosion.

Galvanic Corrosion PDF Corrosion Stainless Steel
Galvanic corrosion describes a process in which two (or more) dissimilar metals are used together, resulting in a corrosive process. A common application that may experience galvanic corrosion is using an attachment, such as a bolt, that is of a different metal than the primary structure, such as a beam. Table of Contents
Galvanic Series (electrochemical series)
A galvanic (voltaic) cell converts the energy released by a spontaneous chemical reaction to electrical energy. An electrolytic cell consumes electrical energy from an external source to drive a nonspontaneous chemical reaction. Example 2.1.1 2.1. 1. A chemist has constructed a galvanic cell consisting of two beakers.

GALVANICCORROSIONTABLE CMP Products Limited
Essentially, galvanic corrosion occurs when two different metals immersed in an electrolyte are joined together. In this scenario, the base or the metal with lesser nobility will undergo corrosion. Thus, the corrosion rate can be determined based on the nobility of metals and the electrolyte to which they're exposed. Advertisement